Supercapacitors combine high energy density, high power density and high cyclability characterisitics to meet energy storage needs that fall in-between that which a battery or capacitor cannot meet. Such needs are prevalent in the transportation, mobile electronics and renewable energy industries which require storage devices of such capability. While the application of supercapacitors is large and sometimes complex, the device itself is relatively simplistic being composed of 4 elementary components: working electrode, counter electrode, electrolyte and separator.
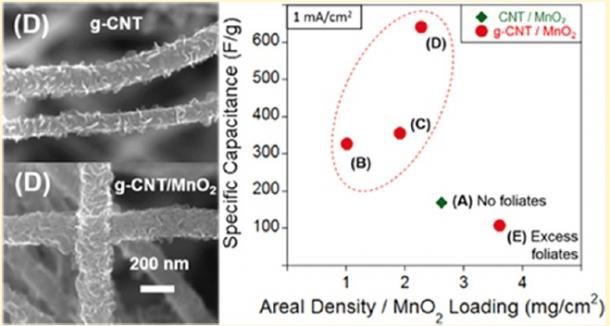
The Nanomaterials and Thin Films Laboratory (NTFL) is primarily interested in studying novel working/counter electrode materials. Such materials have included multi-walled carbon nanotubes (MWCNTs), graphenated carbon nanotubes (g-CNTs) and a variety of these CNTs coated with metal oxides to enhance capacitance and performance. By either manipulating CNT morphology or depositing metal oxides via atomic layer deposition (ALD), electrodeposition (ED) or other processes, NTFL has been able to create tunable and highly capacitive supercapacitor electrodes with high cyclability. Recent efforts have been to coat g-CNTs with MnO¬2 via ED to improve specific capacitance (Fig. 1).
Recent Publications
Brown, B; Cordova, IA; Parker, CB; Stoner, BR; Glass, JT(2015)."Optimization of Active Manganese Oxide Electrodeposits Using Graphenated Carbon Nanotube Electrodes for Supercapacitors" Chemistry of Materials.27(7): 2430- 2438 More info
Ubnoske, SM; Raut, AS; Parker, CB; Glass, JT; Stoner, BR(2015)."Role of nanocrystalline domain size on the electrochemical double-layer capacitance of high edge density carbon nanostructures" MRS Communications.: 1- 6 More info
Stoner, BR; Brown, B; Glass, JT(2014)."Selected topics on the synthesis, properties and applications of multiwalled carbon nanotubes" Diamond and Related Materials.42: 49- 57 More info
Brown, B; Parker, CB; Stoner, BR; Grill, WM; Glass, JT(2012)."Electrochemical charge storage properties of vertically aligned carbon nanotube films: Effects of thermal oxidation" The Journal of Physical Chemistry C.116(36): 19526- 19534 More info
